|
Mg(OH) 2(s) 2HCl (aqueous) - MgCl 2(aqueous) 2H 2O(l) The chemical equation for these reactions areĢKOH(aqueous) CaCl 2(aqueous)-Ca(OH) 2(aqueous) 2KCl(aqueous)ĪgNO 3(aqueous) NaCl(aqueous) - AgCl↓ NaNO 3 (aqueous) The reaction between magnesium hydroxide (Mg(OH) 2) and hydrochloric acid (HCl), resulting in the formation of magnesium chloride(MgCl 2) that is an insoluble salt. 
The reaction between silver nitrate (AgNO 3) and sodium chloride (NaCl), results in the formation of silver chloride (AgCl) that is an insoluble salt. Some examples are reaction between calcium chloride (CaCl 2) and potassium hydroxide(KOH), resulting in the formation of calcium hydroxide that is an insoluble salt. With the help of precipitation reactions, we can determine the presence of different ions present in a particular solution. In the reaction of potassium chloride and silver nitrate, results in the formation of silver chloride(AgCl) which is a white precipitate in a solid-state and insoluble in water. The equation of the reactionĪgNO 3(aqueous) KCl(aqueous) -–AgCl(precipitate) KNO 3(aqueous) The chemical reaction between potassium chloride (KCl) and silver nitrate (AgNO 3), and solid silver chloride (AgCl) is the precipitate or the insoluble salt formed as a product of the reaction is one of the examples of a precipitation reaction. Thus, either its cation or anion starts bonding with another ion in solution. In a single replacement reaction, one ionic compound dissociates. In a double displacement reaction, both ionic reactants dissociate in water and their ions bonds with the respective cation or anion from the other reactant.įor a double displacement reaction to be a precipitation reaction, one of the resulting products must be insoluble in aqueous solution. They can be single displacement reactions or double displacement reactions. These insoluble salts formed are precipitates that are the products of it. It means the chemical reaction occurs in aqueous solutions where two ions bond together to form insoluble salts. Thus, the reactants can be in forms of solid, gas or liquid. Moreover, chemical reactions occur between two or more chemical compounds that we refer to as reactants. Thus, it gives rise to a new element under some particular conditions. Further, chemical reactions consist of chemical changes that take place within the substances. 
of Fe(OH) 3 is obtained.1.6 Solved Question for You Precipitation ReactionĪ precipitation reaction is a chemical reaction that occurs in aqueous solution and form precipitates. (iii) When ammonia solution is added dropwise to iron (III) chloride, a reddish brown ppt. When ammonium salt is heated with caustic soda solution, ammonia gas is evolved.Īmmonium Salt Sodium Hydroxide → Δ \xrightarrow Zn(OH) 2 ( NH 4 ) 2 SO 4 2 NH 4 OH ⟶ zinc (II) sulphate Tetraammine SO 4 4 H 2 O What do you observe when ammonium salt is heated with caustic soda solution? Write the word equation: 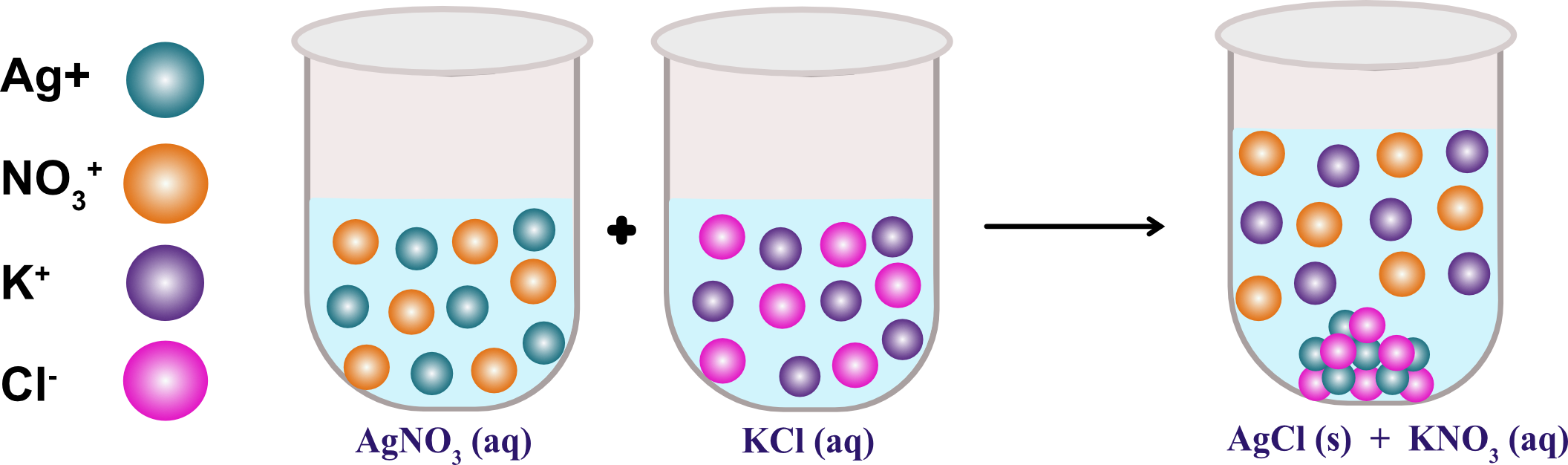
(a) White precipitate insoluble in NH 4OH but soluble in NaOH. Name the probable cation present based on the following observations: (d) Aluminium acetate - Colourless Question 3 Write the probable colour of the following salts: The solid thus formed is called Precipitate. (d) Precipitation - The process of formation of an insoluble solid when solutions are mixed is called Precipitation. (c) Reagent - A reagent is a substance that reacts with another substances. (b) Qualitative analysis - Identification of the unknown substances in a given sample is called Qualitative analysis. (a) Analysis - Determination of the chemical components in a given sample is called Analysis. Chapter 4 Analytical Chemistry Class 10 - Concise Chemistry Selina
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
